The Research Office is managed by Dr Sadhbh O’Neill
The Research Office is being launched to align TUH resources with the TUH Research Strategy and as a response to the needs of researchers within TUH.
The function of the research Office is laid out in the Organogram below:
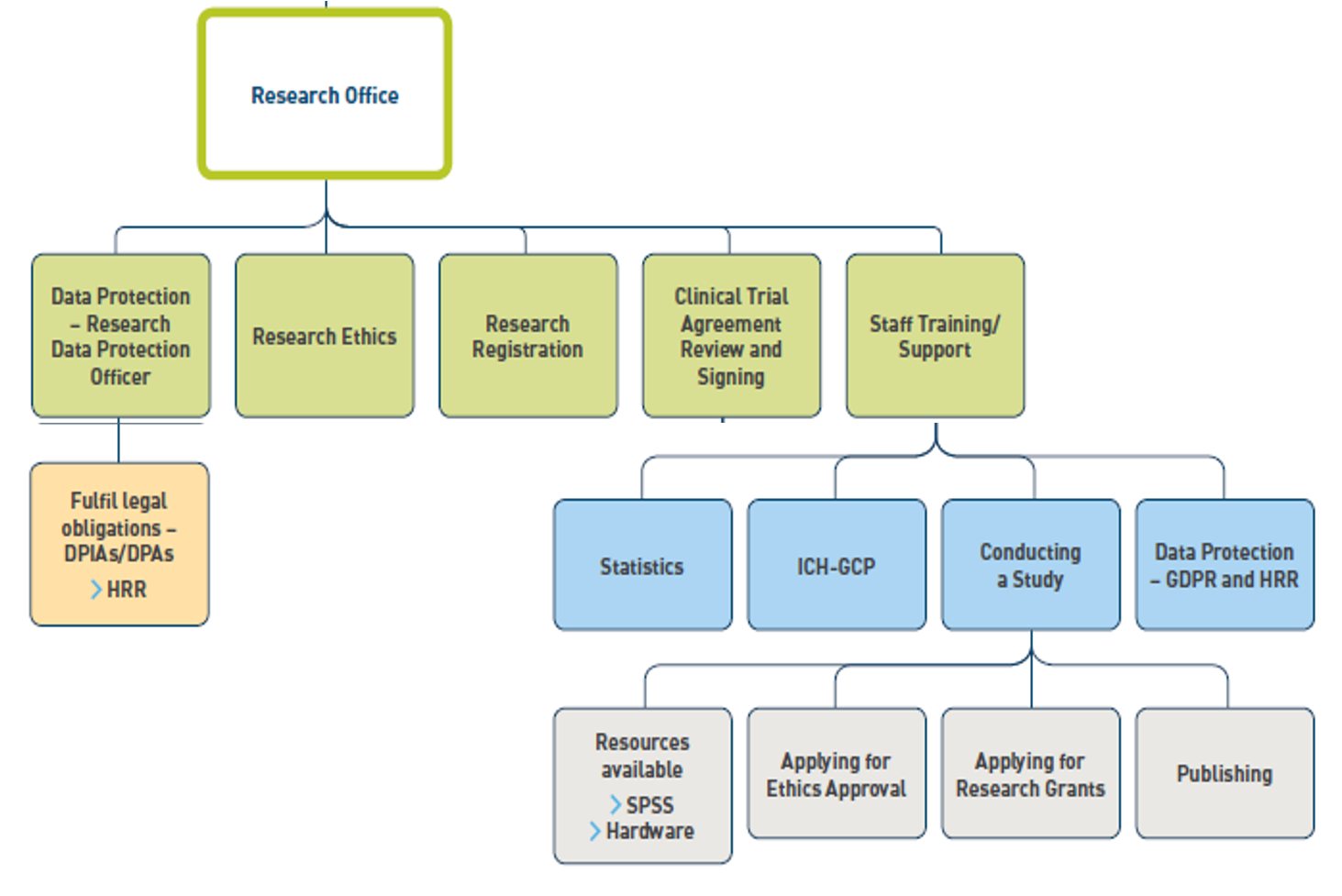
- Research Registration: All research, clinical or other should be registered with the Research Office through the Registration Form. Registering your research will include providing names and affiliations of all researchers involved in the study, financial aspects of the study if applicable, and data protection considerations.
- Research Ethics: All research involving the recruitment of staff or patients must receive ethical approval before beginning by applying through Research Ethics Application Form. Please see the Research Ethics Committee section for further details.
- Data Protection: All research is subject to the Data Protection Legislation. Please see Research Data Protection page
- Clinical Trial and Observational Study Agreement, Material Transfer Agreement and Data Sharing Agreement sign off: The DCEO is the designated signature for the above agreements in TUH. The Clinical Trials Officer, Ms Colette Hoare is responsible for the review of these agreements. Please see the Clinical Trial and Observational Study Agreement Sign Off
- Staff Training, Development and Support: The Office will offer training such as ICH-GCP training to researchers, other training will also be offered. Resources such as SPSS and networked laptops with remote access will be provided through the Research Office.